|
As for the rightmost Carbon, we also have a tetrahedral where Carbon binds with one Carbon and 3 Hydrogens.ĭon’t Miss: What Are Probes In Biology What Is The Difference Between Electron Domain Geometry And Molecular Geometry Use An Example Continuing this trend, we have another tetrahedral with single bonds attached to Hydrogen and Carbon atoms. Again, we have 4 electron groups which result in a tetrahedral. 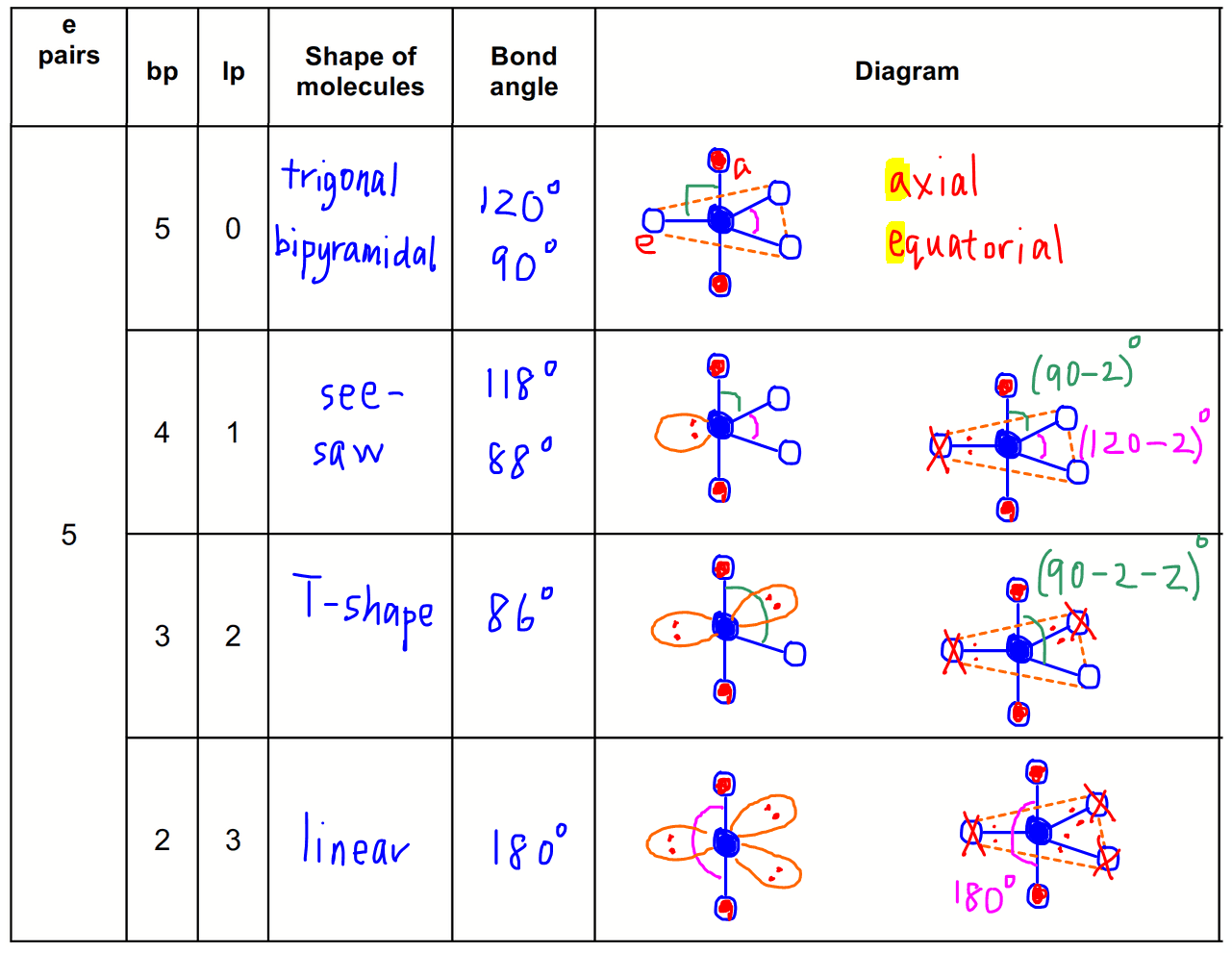
This Carbon has 2 single bonds to 2 Carbons and 2 single bonds to 2 Hydrogens. By checking the geometry of molecules chart above, we have a tetrahedral shape. That means that we have 4 electron groups. We see that C has three single bonds to 2 Hydrogens and one single bond to Carbon. You can view a better structural formula of butane at en./wiki/File:Butane-2D-flat.png If we break down each Carbon, the central atoms, into pieces, we can determine the relative shape of each section. C-C-C-C is the simplified structural formula where the Hydrogens are implied to have single bonds to Carbon. Follow the example provided below:īutane is C4H10. In other words, we take long chain molecules and break it down into pieces. For the final description, we combine the separate description of each atom. We take in account the geometric distribution of the terminal atoms around each central atom. The VSEPR theory not only applies to one central atom, but it applies to molecules with more than one central atom. Molecules With More Than One Central Atom When electrons are bonded to the central atom and do not have lone pairs, the electron geometry and molecular geometry are similar. If the lone pairs of electrons are situated in the molecule, it changes the molecular geometry and not the electron pairs. The definition of electron geometry is the arrangement about electron groups of atoms. Molecular geometry in chemistry is a concept that refers to the arrangement of atoms about the central atom in a three-dimensional space. Whats The Main Difference Between Electron Pair Geometry & Molecular Geometry Looking at the positions of other atomic nuclei around the central determine the molecular geometry.Remember electron groups include not only bonds, but also lone pairs! Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons.To sum up there are four simple steps to apply the VSEPR theory. Molecular Geometry VS Electron Geometry – The Effect of Lone Pairs on Molecular Shape
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
